
Heart rate was monitored each 5 min and oral temperature was measured during each of the nonexercise intervals to ensure that subjects were not overly strained.Īfter the cessation of exercise, subjects voided for collection of urine samples, were weighed, and then entered an adjacent chamber (28☌, <30% rh) for a seated recovery period without any fluid supply. Sweat from the forearm bag was collected every 30 min of exercise the bag for collecting sweat was changed from one forearm to the other in consecutive collection periods to minimize the pore occlusion, which influences excretion rate and electrolyte concentrations in sweat (Sohar et al., 1965). Exercise continued until body weight decreased between 2 and 3%. Exercise (40% )began for an initial 30-min period, followed by alternating 5-min rest and 15 min exercise periods. Subjects then sat in the contour chair of the cycle ergometer in a semirecumbent position and had electrocardiogram electrodes put in place and a vinyl bag for collecting sweat placed on one forearm. Body weight was measured to the nearest 1 g. After a control blood sample (5 ml) was taken, subjects emptied their bladders and entered another environmental chamber (36☌, <30% rh). On the day of the experiment, subjects reported to the laboratory normally hydrated but without breakfast, then entered an environmental chamber and rested in the sitting position for 30 min at a thermoneutral temperature. We hypothesized that a lower sweat would be accompanied by a smaller reduction in plasma water loss during dehydration.Ībout 1-2 weeks before the experiment, maximal aerobic power ( ) (cycle ergometer) and plasma volume were measured on each subject. The purpose of this study was to clarify the effect of sweat on water mobilization from the ICF compartment in conditions of thermal stress. However, there is no experimental evidence that shows a relation between and water mobilization from the ICF space. It is well known that sweat decreases during the process of heat acclimation (Kirby and Convertino, 1986 Locke et al., 1951). These results suggest the necessity of measuring electrolyte balance to analyze water balance between fluid compartments. Water movement might be linked to the electrolyte losses from each compartment, as suggested by Nose et al. (1986) reported that in rats a higher water loss occurred from the extracellular fluid (ECF) space than from the ICF space during dehydration of 10% body wt. There would then be less mobilization of water from the intracellular fluid (ICF) space due to the smaller increase of extracellular osmolality. Costill (1977) ascribed the relatively greater plasma water loss to movement accompanying the major ions lost in sweat and urine, which are those of the extracellular compartment. Senay (1979) recently reviewed the dehydration literature and reported that water appeared to be lost from the plasma at a rate one to five times that of other fluid compartments during dehydration. Among the possible compensations is the body's ability to mobilize water from the extravascular to the intravascular space (Miki et al., 1983b Mohsenin and Gonzalez, 1984 Morimoto et al., 1981 Nose et al., 1983). A reduction of the central circulating blood volume, due either to hypovolemia accompanying dehydration or dilation of the peripheral vasculature, results in a fall in cardiac filling pressure and stroke volume and, if uncompensated, also in cardiac output (Fortney et al., 1983 Miki et al., 1983a). The cephalic and basilic veins are connected at the elbow by the median cubital vein.Maintenance of blood volume is important for optimal regulation of both arterial blood pressure and body temperature during exercise and thermal stress (Fortney et al., 1981a,b Fortney et al., 1983 Nadel, 1984). Within the axilla, the cephalic vein empties into axillary vein. It ascends the antero-lateral aspect of the upper limb, passing anteriorly at the elbow.Īt the shoulder, the cephalic vein travels between the deltoid and pectoralis major muscles (known as the deltopectoral groove), and enters the axilla region via the clavipectoral triangle. 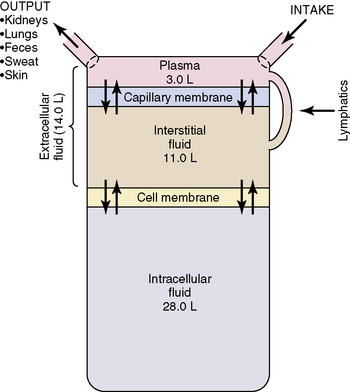
The cephalic vein also arises from the dorsal venous network of the hand. Here, it combines with the brachial veins from the deep venous system to form the axillary vein. The basilic vein originates from the dorsal venous network of the hand and ascends the medial aspect of the upper limb.Īt the border of the teres major, the vein moves deep into the arm. They are located within the subcutaneous tissue of the upper limb. The major superficial veins of the upper limb are the cephalic and basilic veins. In this article, we shall look at the anatomy of the upper limb veins - their anatomical course, structure, and their clinical relevance. It can be subdivided into the superficial system and the deep system. The venous system of the upper limb drains deoxygenated blood from the arm, forearm and hand.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
